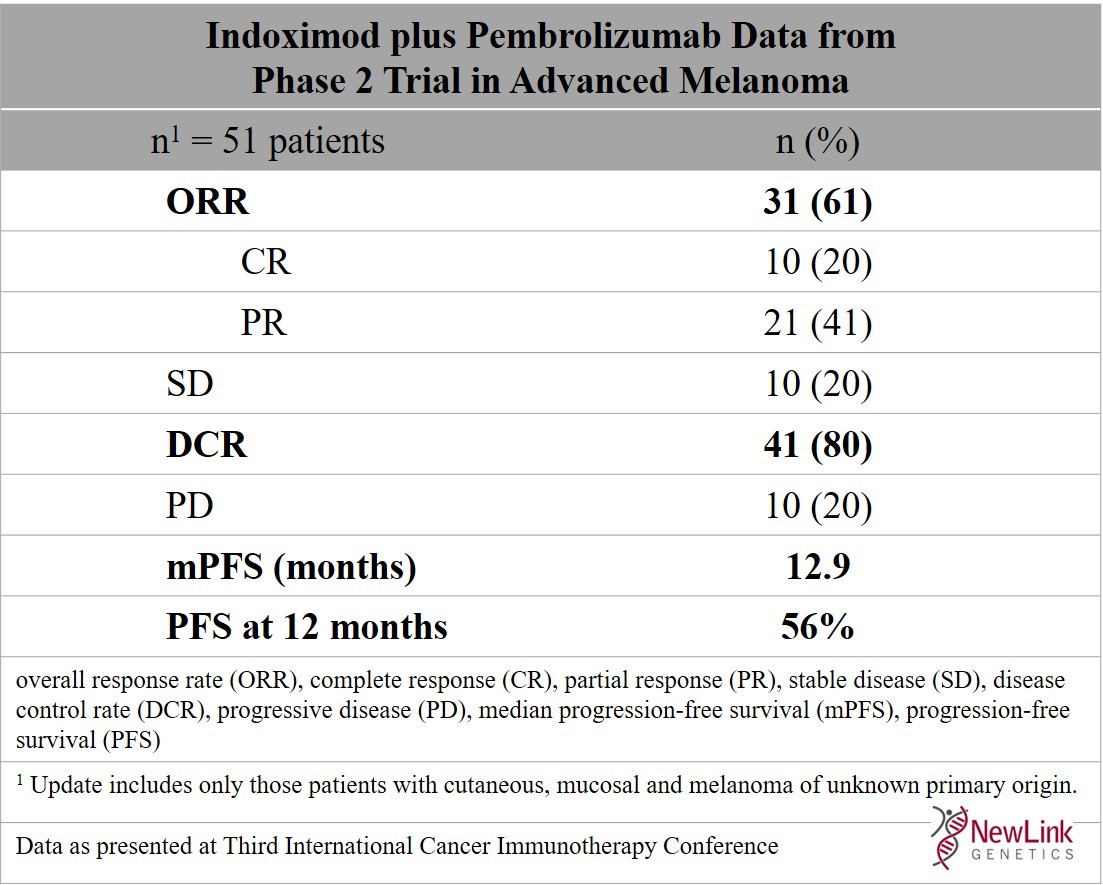
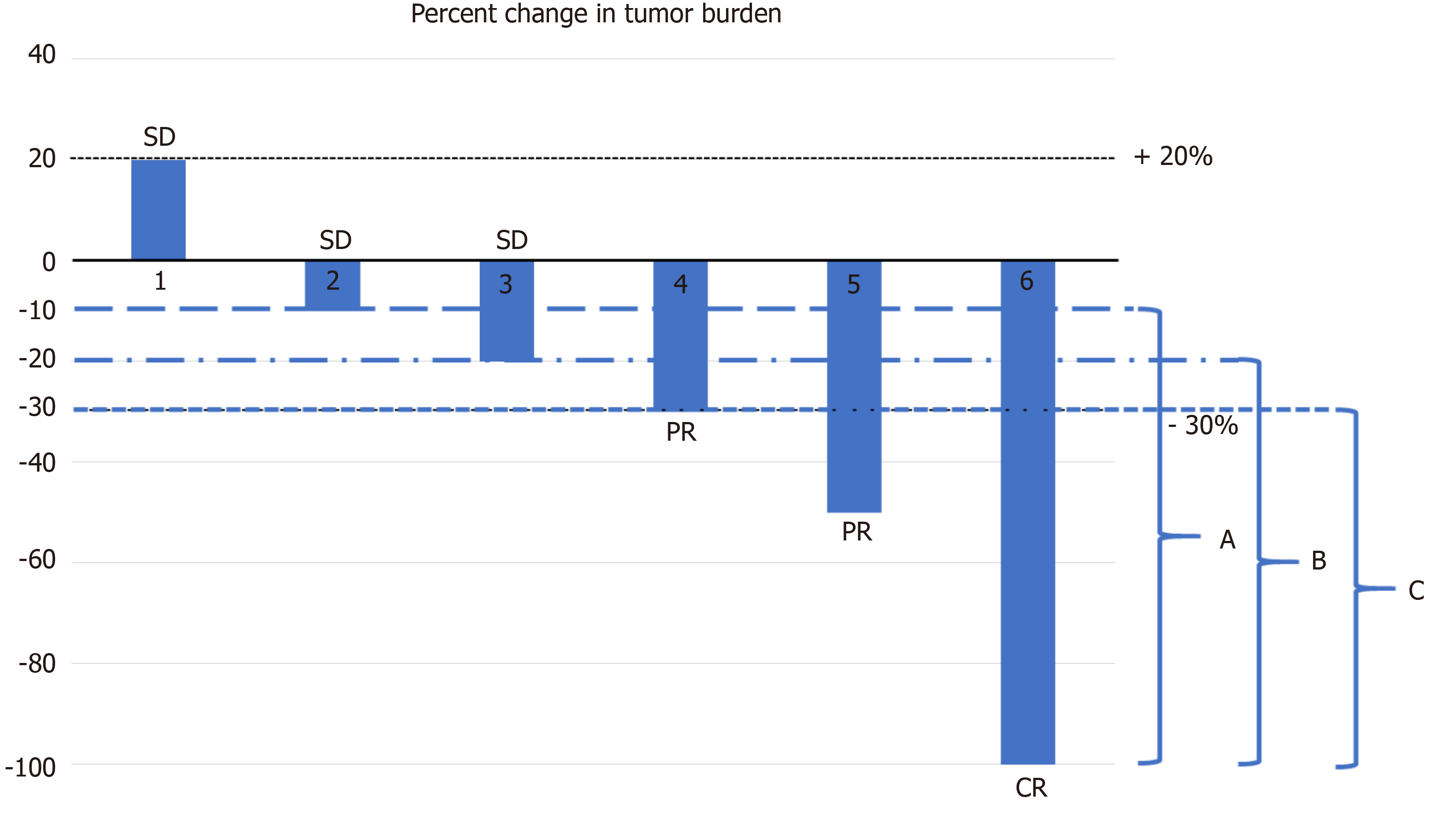
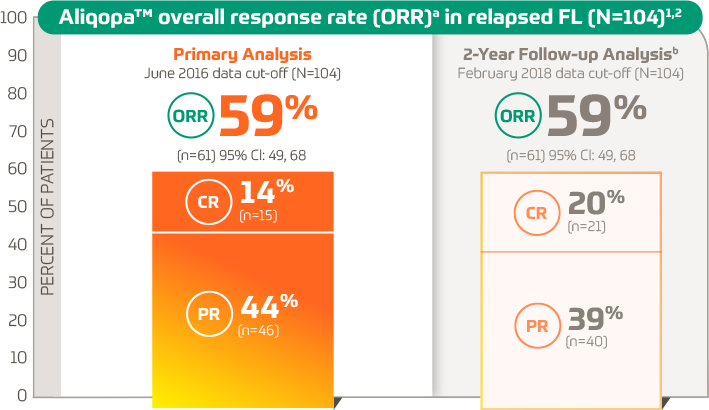
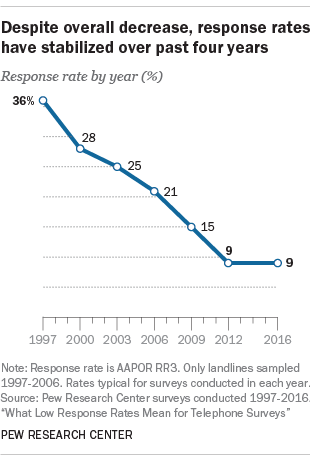
Objective response rate of placebo in randomized controlled trials of anticancer medicines - eClinicalMedicine
Nexcella Announces Positive 58-Patient NXC- 201 Clinical Data: 100% Overall Response Rate in light chain (AL) Amyloidosis; 92% Overall Response Rate in Multiple Myeloma at the EBMT 49th Annual Meeting in Paris | BioSpace
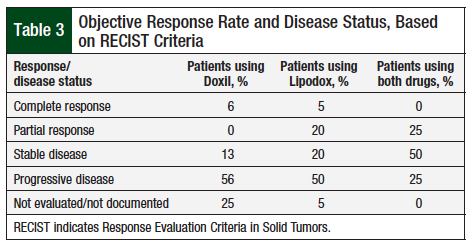
Comparing Outcomes in Patients with Recurrent or Refractory Ovarian Cancer Managed with 1 of 2 Versions of Pegylated Liposomal Doxorubicin at an Academic Medical Center

Predicting Successful Phase Advancement and Regulatory Approval in Multiple Myeloma From Phase I Overall Response Rates | JCO Clinical Cancer Informatics
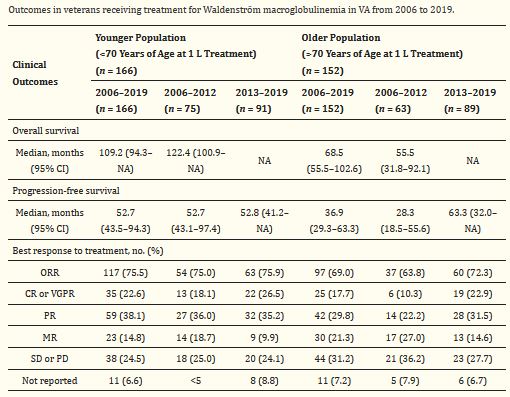
Response Rates for Waldenström Macroglobulinemia Improve With Use of New First-Line, Better-Tolerated Treatments - U.S. Medicine

Overall Response Rate in Previously Untreated CLL | CALQUENCE® (acalabrutinib) 100 mg tablets | For HCPs